MDCG 2021‑24 Rev.1 — What is changing in the classification of medical devices?
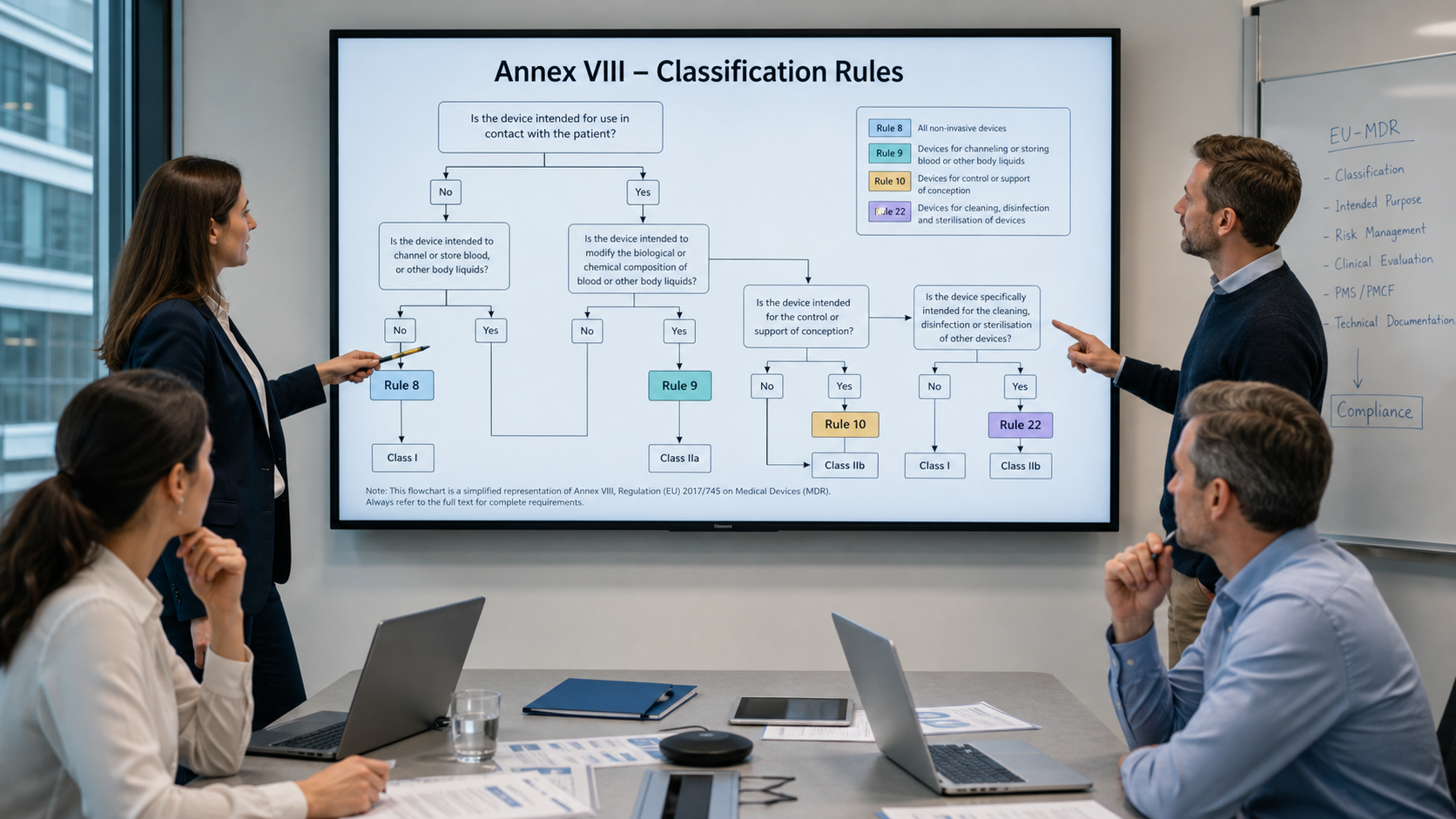
1. Background and objectives
Guideline MDCG 2021‑24 helps manufacturers, notified bodies and other actors to apply the classification rules set out in Annex VIII to Regulation (EU) 2017/745 (MDR) consistently. Revision 1 (MDCG 2021‑24 Rev.1) published in April 2026 clarifies the interpretation of these rules in several places and supplements practical information on typical borderline cases.
The classification of a device in Classes I, IIa, IIb or III is central to the further conformity assessment process, clinical requirements and post-market effort. Classification errors can lead to delays in the approval process or to complaints from notified bodies and authorities.
2. Relevant changes in MDCG 2021‑24 Rev.1
The revision focuses primarily on clarifications and on refining practice-relevant examples. The following are particularly affected:
- the definition of implantable devices and the term clinical intervention
- the language version for applying the classification rules (focus on “product” instead of “device” only)
- Tables and practical information on Rules 2, 8, 9, 10, 12, 16 and 22
For daily practice, this means in particular:
- a more precise differentiation of product groups from the respective risk classes
- additional explanations of borderline cases, in particular for implantable, active and complex systems
- a clearer integration of Annex XVI products into the classification logic
3. Overview of the changed content
The following table summarizes the main substantive adjustments to Revision 1. It is suitable as a starting point for an internal review of existing classification decisions.
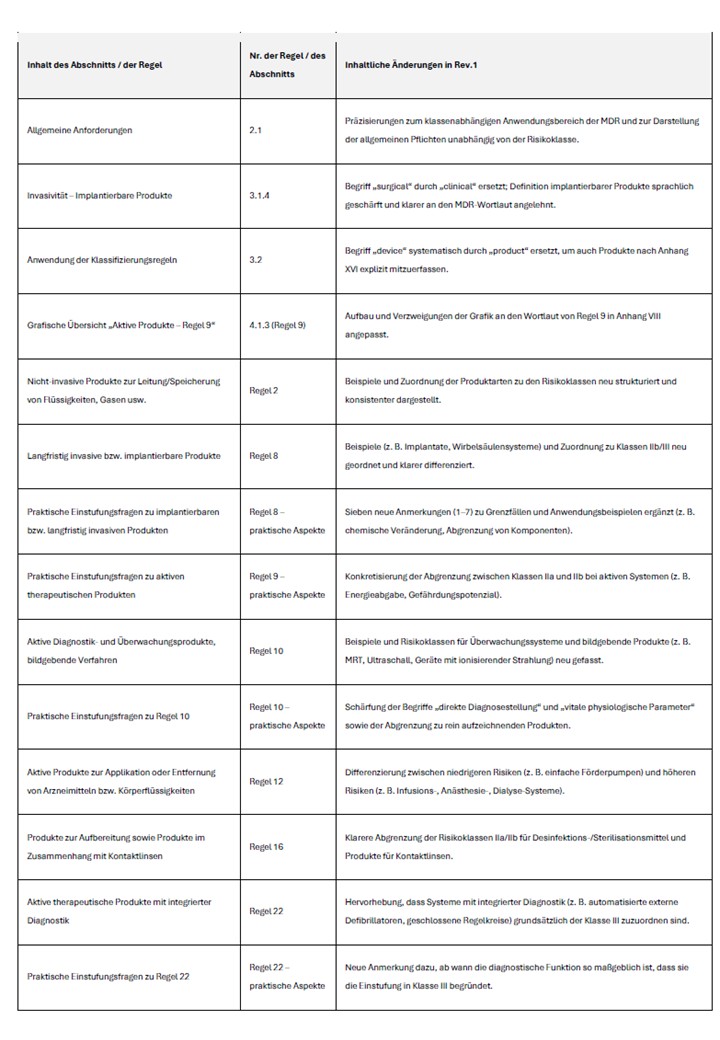
4. Recommendations for manufacturers and notified bodies
For manufacturers of medical devices and Annex XVI devices, it is advisable to specifically review the existing classification in the following cases:
- Products with implantable components or long-term invasive use
- active therapeutic systems with significant energy output
- active diagnostic and monitoring devices, particularly when making a direct diagnosis
- complex systems with integrated diagnostics (e.g. closed loop systems, AEDs)
Specifically, at least the following documents and processes should be adapted by the company:
- Classification guidelines and work instructions (SOPs)
- Training materials for regulatory affairs, development and clinical teams
- internal decision-making tools (e.g. classification matrices, tools, checklists)
sources
Medical Device Coordination Group (MDCG): Guidance on classification of medical devices. MDCG 2021‑24, version 2021.
Medical Device Coordination Group (MDCG): Guidance on classification of medical devices. MDCG 2021‑24 Rev.1, April 2026.
For researching companies, the more precise MDCG guidance provides the opportunity to align classification strategies, study designs and clinical development plans more specifically with MDR requirements right from the start and thus make approval processes more predictable and efficient.
Is that interesting for you? Contact Feel free to contact us for an initial consultation!
Show the medical benefits of your product
With our many years of experience and expertise, we offer effective solutions to demonstrate the medical benefits of your product.
From the conception to the execution of preclinical and clinical investigations, we support you with customized services.
Find out how MEDIACC can help you achieve reimbursability for your products.
